RESPECT patient needs
RESPECT: Relating Expectations and Needs to the Participation and Empowerment of Children in Clinical Trials
Project overview
Studies have shown that over 50% of the medicinal products used in children may not have been tested or authorised for use in this age group. This leaves health care professionals with no alternative but to use medicines "off-label", judging the suitability and the correct dose of these medicines themselves in the absence of paediatric labeling information. This poses significant risks of inefficacy and/or adverse reactions for children.
The EU regulation (EC No. 1901/2006 on Medicinal Products for Paediatric Use) came into effect in January 2007 to ensure that medicinal products that are researched, developed and authorised in Europe will also meet the therapeutic needs of children. This will mean an increased demand for children to participate in clinical trials of new medicines.
The European research project RESPECT, led by the Institute of Clinical Sciences at the Sahlgrenska Academy at Gothenburg University, Sweden, set out to identify the needs and motivations of children and their families who have participated or might participate in clinical trials in Europe.
We explored the issues raised by children's participation in medical research by speaking with paediatric patients and their parents, patient support organisations, paediatricians, ethical committees and representatives of the pharmaceutical industry to gather their experience, insights and suggestions. This enabled us to make recommendations for how to empower children such that medical researchers respect their interests and can motivate their participation in future clinical trials.
Please see the Publications below for our presentations and articles.
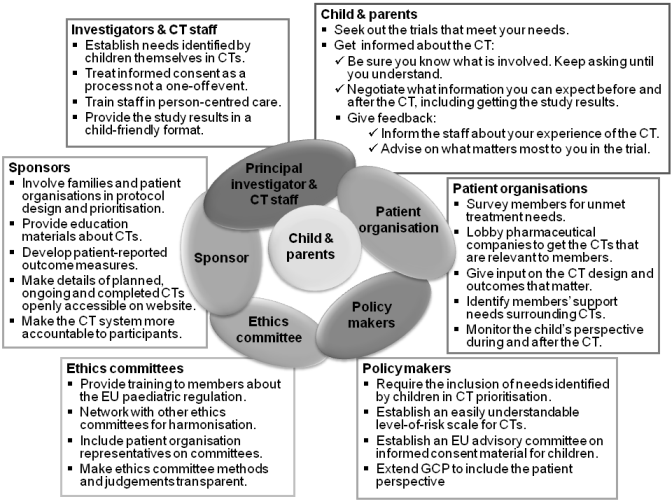
RESPECT: Main recommendations to stakeholders
A full description and discussion of these recommendations can be found in chapter 9 of the RESPECT book.
RESPECT project dissemination meeting presentations (Brussels, 25 May 2011)
| Authors | Title |
|---|---|
| John Chaplin | Overview of the RESPECT project and the paediatric clinical trials landscape |
| Adriana Ceci | View from the Paediatric Committee of the EMA |
| Catriona Chaplin | Young patients’ and their parents’ experiences of clinical trials |
| Pia-Sophie Wool | Recruiting children to paediatric clinical trials – the view from the clinician |
| Annagrazia Altavilla | Involving children in decision-making – what are their rights? |
| Falk Wulf | How do families make the decision to participate? |
| Monika Bullinger | Patient-reported outcomes - what matters to the child? |
| Cristina Manfredi | Clinical trial networks – how can the patient or parent play an active part? |
| Liuska Sanna | What do patient organisations want their role to be? |
| Francis P. Crawley | Promoting transparency in European paediatric clinical research |
| (all) | Breakout workshop reports |
Conference presentations
| Authors | Title | Conference | Date |
|---|---|---|---|
| John Chaplin & the RESPECT project | How to improve recruitment and retention in paediatric clinical trials | Swedish Society of Medicine (SVLS) Annual conference, Stockholm | 30 Nov - 1 Dec 2011 |
| Annagrazia Altavilla | Research with children: current trends and perspectives | International Symposium: Convention on Human Rights and Biomedicine - updated or outdated?, Coimbra, Portugal | 12 July 2011 |
| Chaplin, Neubauer, Wulf, v. Mackensen, Sanna & Chaplin | Mutual Respect and Shared Goals for Clinical Trials on Children | 1st Global Congress for Consensus in Pediatrics & Child Health (CIP), Paris | 17-20 Feb 2011 |
| Wulf, Chaplin, Sanna, Neubauer, Crawley, Giaquinto, Ceci, Bullinger & Chaplin | RESPECT: Good practice in empowering families in clinical trials | PatientPartner final workshop: Patients Partnering in Clinical Trials, Brussels | 7-8 Dec 2010 |
| Chaplin, Chaplin, Pfeiffer-Mosesson & the RESPECT project | Securing children’s participation in clinical trials | Swedish Society of Medicine (SVLS) Annual conference, Gothenburg | 1-3 Dec 2010 |
| Sandberg, Chaplin & Chaplin | Beyond Informed Consent: Educating parents to empower them in the clinical trial process | Swedish Society of Medicine (SVLS) Annual conference, Gothenburg | 1-3 Dec 2010 |
| Chaplin, Pfeiffer-Mosesson & Chaplin | RESPECT project: Why do parents let their children participate in clinical trials? | Swedish pharmacists annual conference: Läkemedelskongressen, Stockholm | 20-21 Oct 2009 |
Publications
The RESPECT consortium consisted of experts from the fields of clinical research, patient representation, and European paediatric research ethics and regulation. The following experts were partners in the RESPECT project:
- John Chaplin PhD , University of Gothenburg, Göteborg Pediatric Growth Research Center (GPGRC)
- The GPGRC is the coordinator of the RESPECT project and has expertise in the fields of patient-reported outcome measures, psychoendocrinology and paediatric clinical trials.
- Francis Crawley ( ) - Good Clinical Practice Alliance (Europe)
- Good Clinical Practice Alliance (Europe) is a consultancy group active in the area of ethics as related to CTs and medical research throughout Europe.
- Liuska Sanna, European Patients Forum
- The EPF is a European umbrella NGO representing 44 chronic disease-specific patient organisations. The EPF works at the European level and with national coalitions of patients' organisations. EPF was the leading coordinator of the Value+ project promoting patients involvement in EU supported health-realted projects.
- Prof Dr Adriana Ceci, Consorzio valutazione Biologiche e Farmacologiche (Pavia)
- Consorzio per le Valutazioni Biologiche e Farmacologiche is a clinical research organisation conducting and monitoring CTs, particularly with Thalassaemia patients, and coordinator of the TEDDY European Network of Excellence.
- Prof Dr David Neubauer, University Children's Hospital Ljubljana and the Foundation of Child Neurology
- The UCHL has expertise in the field of conducting paediatric clinical trials, particularly children with epilepsy and mitochondrial disease.
- Prof Dr Carlo Giaquinto, Azienda Ospedaliera di Padova
- Azienda Ospedaliera di Padova - Dept of Pediatrics, University of Padua has expertise in the area of paediatric CTs with a focus on HIV. They are leading the EU project GRIP and led the steering committee of the PENTA paediatric CTs network.
- Prof. Dr Monika Bullinger,University Hospital of Hamburg-Eppendorf, Department of Medical Psychology
- University Medical Centre Hamburg-Eppendorf has expertise in the areas of patient-reported outcome measures and the development of quality-of-life instruments for child self report (eg. (DISABKIDS). The Medical Psychology Dept. trains medical staff who will in the future conduct CTs with children.


